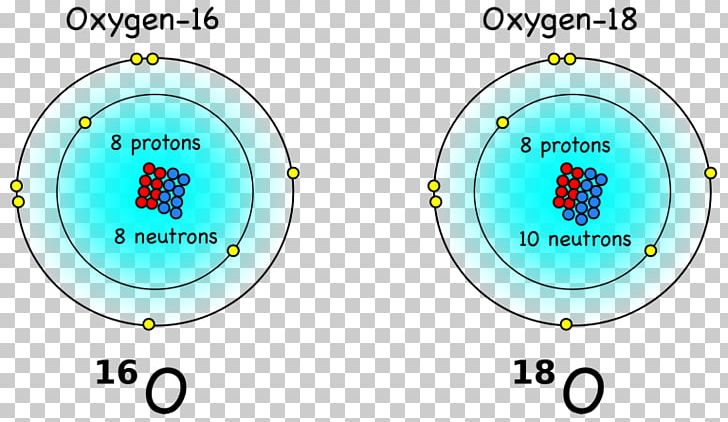
Therefore, there are various non-equivalent definitions of atomic radius. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. Because neutral atoms in the periodic table have the same number of electrons as protons, the atom must have 8 protons. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Oxygen atom is 66pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Oxygen are 16 17 18. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The number eight also means that oxygen has eight protons. In writing the electron configuration for oxygen the first two electrons will go in the 1s orbital. Oxygen with the symbol O has the atomic number 8 which means it is the 8th element in the table. Oxygen forms a chemical compound (O2) of two atoms which is a. Oxygen is the eighth element with a total of 8 electrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Oxygen is a chemical element with an atomic number of 8 (it has eight protons in its nucleus). Thus, what is the atomic mass of an oxygen atom. Oxygen has an atomic number of 8 and, most commonly, a mass number of 16. Which 4 of these 25 elements make up approximately 96 of living matter. Neutron number plus atomic number equals atomic mass number: N+Z=A. to lose electrons for the reaction with chlorine or oxygen Lithium has a electron configuration of 2,1 and potassium 2,8,8,1 ( higher atomic number). Terms in this set (23) About 25 of the 92 natural elements are known to be essential to life. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Oxygen is a chemical element with atomic number 8 which means there are 8 protons in its nucleus. 
The nuclear symbols are written this way (again, pretend the superscript and subscript are sitting right on top of each other beside the element symbol): 168 O, 178 O, 188 O. The mass numbers for oxygen must be 8 + 8 16 8 + 9 17 8 + 10 18. You will need to refer to a periodic table for proton values.Atomic Number – Protons, Electrons and Neutrons in Oxygen The element symbol for oxygen is O and its atomic number is 8. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. Thus, we use O2 as the formula for oxygen, N2 for nitrogen, and so on. 
For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Those nonmetals that occur as diatomic (two-atom)molecules are listed in Table 3.6. The "A" value is written as a superscript while the "Z" value is written as a subscript. /oxygen--illustration-545864495-57013d0d5f9b58619532f03c.jpg)
Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
